Clinical history:
A one year-old, female horse presented with weight loss and diarrhoea. She was euthanased, but was one of four horses that had died in the last 2 months on the same property (with a history of Salmonella, Cryptosporidium and Coronavirus on the farm).
On post mortem examination pinpoint lesions were observed on the intestinal lining. Findings were otherwise normal. Multiple fixed tissues were submitted to the laboratory for histopathology examination.
Histopathology results:
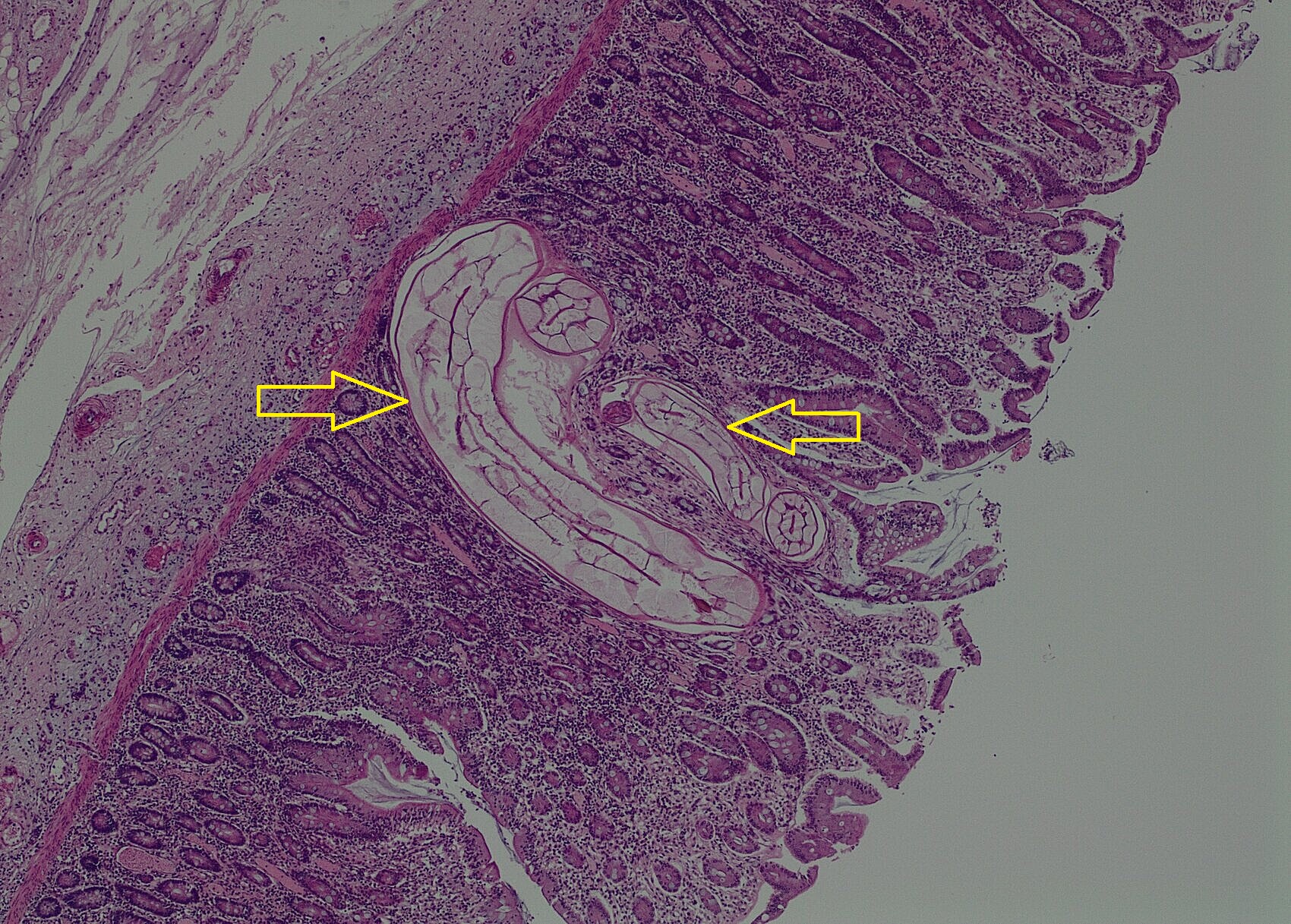
Caecum – Multifocally the mucosa contains moderate numbers of nematode larvae in the lamina propria surrounded moderate numbers of eosinophils, plasma cells and lymphocytes. In some areas there are also small infiltrates of neutrophils, short bacilli bacteria and deposits of fibrin.
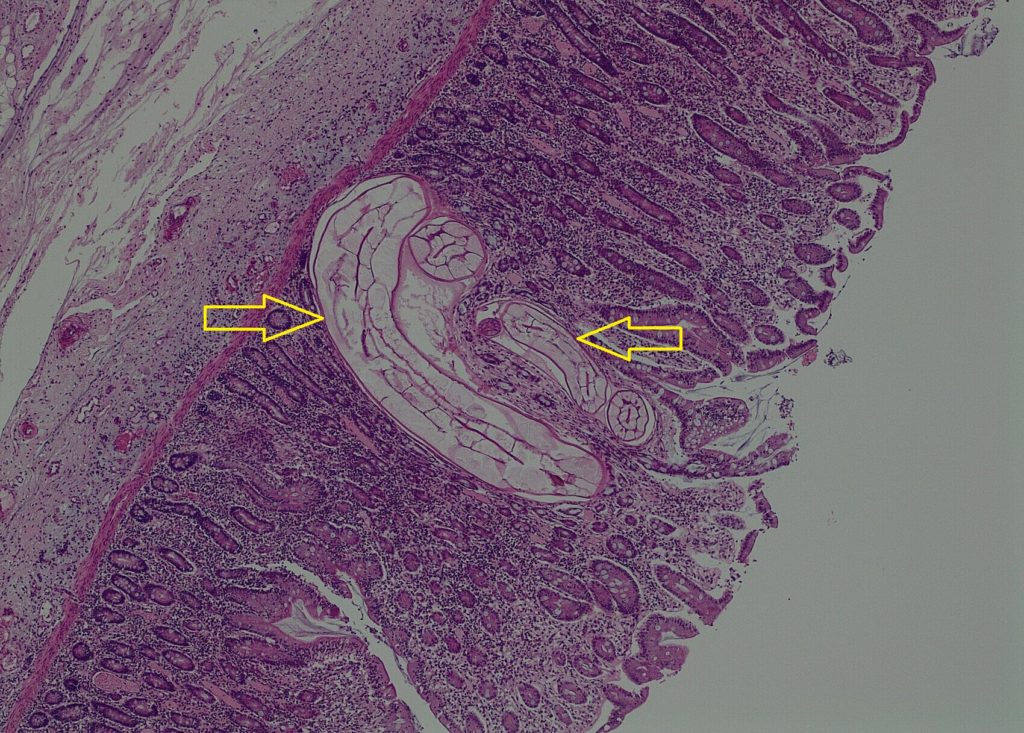
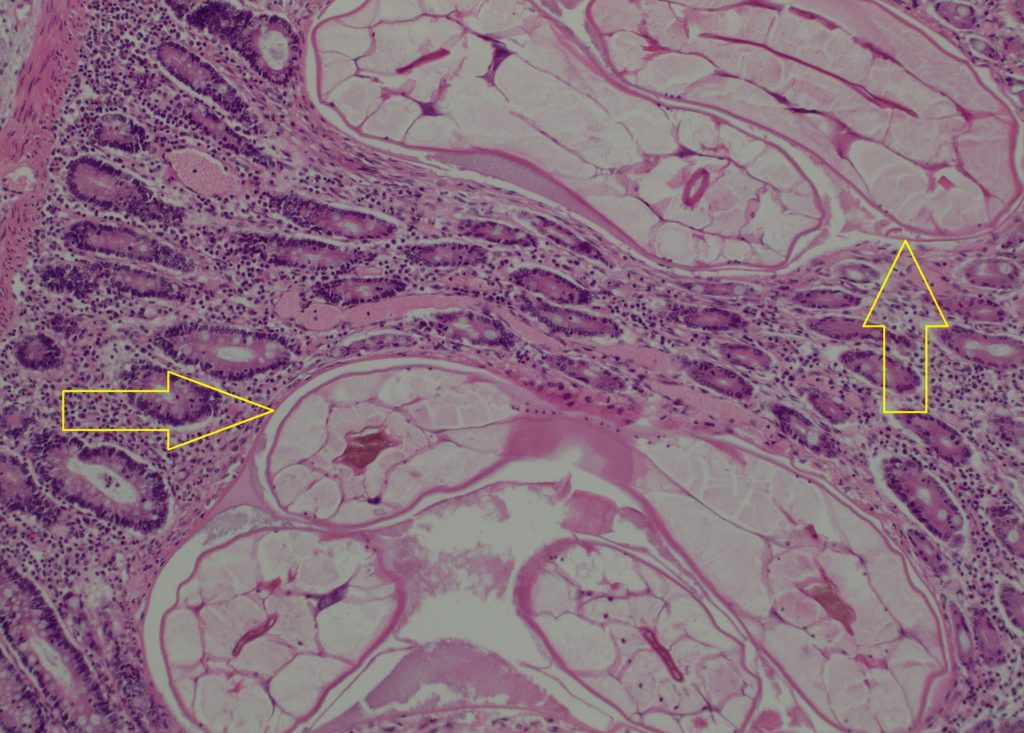
Diagnosis: Caecum – (Cyathostominosis) colitis with mucosal larval small strongyles (cyathostomins /cyathostomes) and neutrophilic colitis with short bacilli bacteria.
Discussion: The likely cause of death was the colitis with acute short bacilli bacterial infection (likely Salmonella, but culture was not performed for identification), but there was also a significant underling chronic-active Cyathostominosis, which likely predisposed the horse to the bacterial infection.
Subacute to chronic diarrhoea in horses almost always involves the large intestine, with or without concomitant small bowel involvement. Salmonella and Clostridium difficile typhlocolitis must be suspected in such cases. Extensive mucosal involvement by larval cyathostomes and strongyles and rarely, ulcerative typhlitis resulting from anoplocephelid tapeworms, may also cause chronic diarrhoea and wasting. Co-infection by Listeria monocytogenes, Salmonella and cyathostomes probably leads to granulomatous typhlocolitis in the horse.
Small strongyles are essentially non-pathogenic as adults. Cyathostominosis is a disease of horses greater than one year of age and little resistance is apparent to repeated infection. The clinical syndrome larval cyathostominosis occurs as a result of simultaneous emergence of inhabited third stage larvae from the intestinal mucosa, and is a significant cause of morbidity and mortality in horses.
The cyathostomins have a direct life-cycle. Infective third stage larval cyathostomins are ingested, and they migrate into the deep mucosa or submucosa of the caecum and colon to encyst and moult, before emerging to the lumen to moult again and mature into adults. Encysted third or fourth stage larvae may undergo hypobiosis or developmental inhibition, persisting in nodules in the colonic wall for as long as two years. The timing during which inhibition occurs is dependent on the climate. Inhibition occurs during colder months of the year in temperate climates, and during the hot summer in tropical climates.
The most devastating damage occurs when large numbers of encysted inhabited larvae emerge en-masse to continue their development in the intestinal lumen. This occurs in the late winter, spring and early summer in temperate climates. Development of widespread in anthelmintic resistance by cyathostomins, particularly encysted larval stages, is well documented. Affected horses may be of any age; clinical signs are non-specific and include diarrhoea, oedema, anorexia and weight loss.
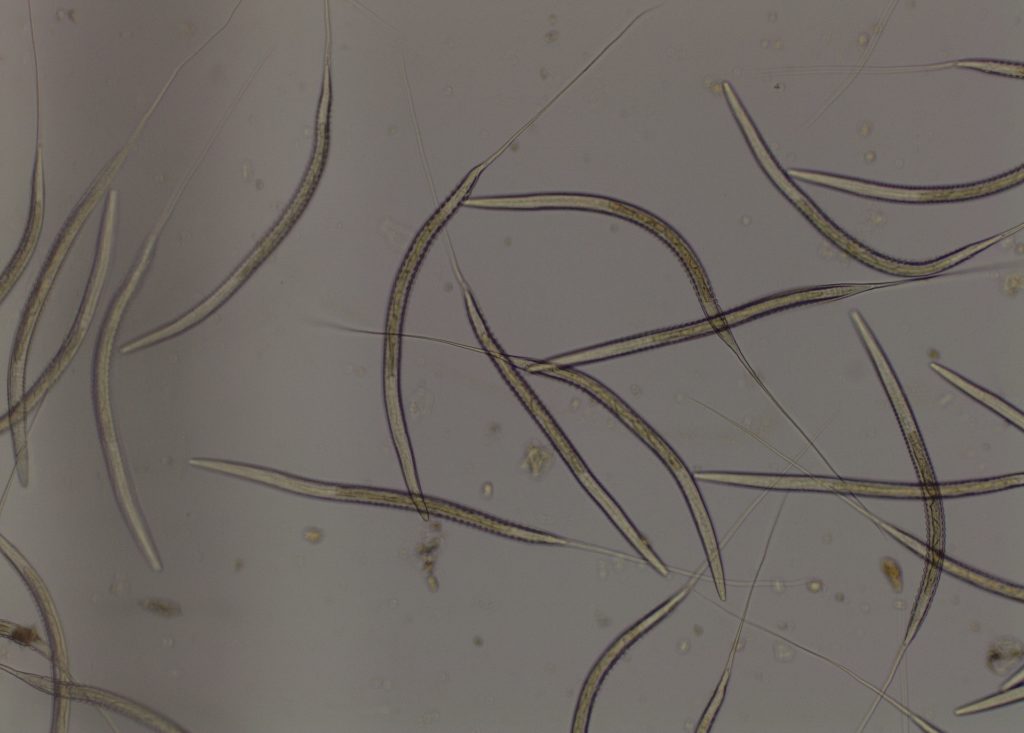
Subsequently, 37 horses from the property had faeces tested for FEC and Cyathostome 4/5th stage larva. Some horses had up to 1200 strongyle eggs/g. Larval culture of a pooled sample of the faeces had 100% Cyathostome larvae.
Due to the sensitivity of FEC testing (only using a small amount of faeces compared to the total faecal output), if no eggs are counted in the FEC it is still possible for eggs to be present (false negative). In this case, if clinical signs indicate cyathostomiasis then larval culture is a more sensitive test to diagnosis Cyathostome infection compared to FEC or Cyathostome 4/5th stage larval tests.
Cyathostome parasitology tests available:
· FEC – Strongyle egg count (Does not differentiate large or small strongyles; may be negative depending on stage of infection)
· Cyathostomes – 4/5th stage larvae (May be negative depending on stage of infection)
· Larval culture – hatches any Strongyle eggs to third stage infective larvae, identifying to genus level and performing a differential of the larvae cultured.
Reference: Jubb, Kennedy and Palmer’s Pathology of Domestic Animals, Sixth Edn, 2015.